CHARLIE (V.O.)
Entropy. A measure of randomness, a
parameter of disorder... energy
broken down in irretrievable heat.
What might appear to be chaos... even
decay... is really a system's way of
smoothing out differences -- its
search for equilibrium. Uncorrelated
parts interact... find
their connections in an evolving
system...... so, from one perspective,
entropy is a clock... charting the
irreversible.
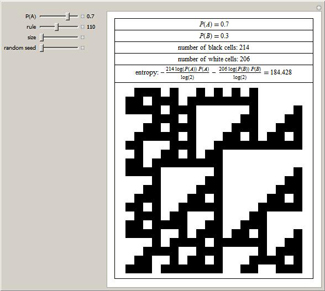
The entropy of a list Q measures its amount of disorder. The initial condition is a finite list of random bits. The
entropy can be used to study the amount of information in the evolution of a
cellular automaton; it is lower in
ordered systems and higher in chaotic (disordered) systems.
Charlie is reading a newspaper as Amita and LARRY come in,
bringing coffee --
AMITA
Thought you snuck out early to work
on that lecture...
CHARLIE
I made the mistake of buying a paper
with my coffee. Remember that idea I
had for a finding in Complex
Polynomial encryption? Withers'
group just patented it.
AMITA
It wasn't Withers... it was his
collective. In fact, a civil engineer
suggested attacking it through
Riemann's Hypothesis.
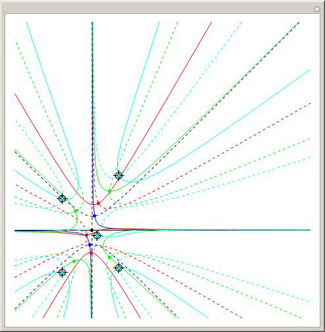
Four points in the
complex plane can be the roots of a complex polynomial of degree four. Solid lines indicate where the real part is zero and dashed lines indicate where the imaginary part is zero. These lines intersect at the chosen roots. The successive derivatives of the complex polynomial behave similarly.
DAVID
(thinking)
127 rolls of floss -- which is a
restricted item in prison for exactly
this reason --
CHARLIE
We might be able to apply a Simplex
Algorithm -- the amount of time it
would have taken to build the ladder,
based on difficulty of access --
LARRY
-- they had limited time to work on
it, when guards and other prisoners
weren't watching them --
AMITA
-- they would have had to find a way
to hide the empty containers, throw
them out...
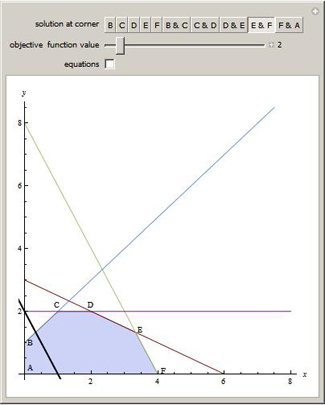
This Demonstration illustrates the graphical solution to several
linear programming problems, all of which have the same set of constraints; you can vary the objective function. When two corner points are optimal, so are all the points on the line segment connecting them. The region shaded in blue is the feasible region and the colored lines correspond to the constraints. The black line represents the chosen objective function set to the slider value.
Larry and Amita give him a "no sale" look --
CHARLIE (cont'd)
Okay... I was seven years old, and I
asked my dad to help me figure out a
good estimate for the remainder term
in a Taylor expansion of the
hyperbolic cosine. I remember our
eyes meeting, and this... tacit
understanding that we'd crossed the
Rubicon.
(beat)
When you're seven, your father needs
to be seven feet tall and infallible.
So I separated math from my Dad...
irrational as it may be, I'd prefer
to keep it that way.
As Larry and Amita exchange a look...
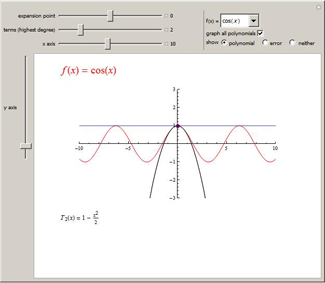
Choose the maximum degree of the
Taylor polynomial to use to approximate a function. You can choose from a variety of functions and manipulate the expansion point. To see the error in the approximation, select the "error" radio button and use the slider that appears under the graph.
AMITA
(exasperated)
Yet again, male communication tests
the limits of Shannon's source coding
theorem.
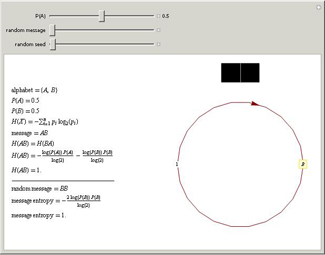
Using the second law of thermodynamics, it is possible to use random variables to calculate the information entropy (or
Shannon entropy) of a message, which is a measure of the amount of information in the message. The probabilities that A
and B occur in the message are P(A) and P(B).
CHARLIE
Sure... where's Maxwell's Demon when
you need him.
ALAN
Maxwell's Demon --
CHARLIE
You know -- the man who stands alone at the
door. In two adjoining rooms, the
temperature and pressure are the
same... a state of perfect equilibrium.
Every time the Demon opens the door,
he admits only those molecules he
chooses... heating one room and
cooling the other... in violation of the
Second Law of Thermodynamics.

Thermal energy is the energy of an object due to random motions of its atoms and molecules. The hotter the object, the
greater its thermal energy. Thermal energy is an extensive variable, proportional to the size of the object. The
individual molecules can have different kinetic energies, but a hot object has a higher average value. In a gas or a
liquid, molecules can move freely in all directions; in a solid, molecules execute small vibrations in all directions
about fixed positions.